
Therefore, this structure is the stable Lewis structure of CF 4. And the outside atoms (fluorines) also form an octet. The electron geometry of ClF3 is Trigonal bipyramidal. The total valence electron present available for drawing the ClF3 Lewis structure is 28. The molecular geometry or shape of ClF3 is T-shaped. In the BI3 Lewis structure, a total of 18 nonbonding electrons and. Two types of bond angles are found in ClF3 87.5 and 175. In the above structure, you can see that the central atom (carbon) forms an octet. Molecular geometry is the name of the geometry used to describe the shape of a molecule. Here, both carbon and fluorine atoms do not have charges, so no need to mark the charges. In the case of bond formation in Acetylene molecule: Name the type of Hybridization. While the Tetrafluoromethane formula and name suggests the CF4 molecular geometry to be tetragonal, the Lewis structure for CF4 further reflects its. Use the following formula to calculate the formal charges on atoms:įormal charge = valence electrons – nonbonding electrons – ½ bonding electronsįor carbon atom, formal charge = 4 – 0 – ½ (8) = 0įor each fluorine atom, formal charge = 7 – 6 – ½ (2) = 0 Explain geometry of methane molecule on the basis of Hybridization. Lone pairs marked, and got the stable Lewis structure of CF 4 #3 Indicate formal charges on the atoms, if necessary Therefore, place carbon in the center and fluorines on either side. Since carbon is less electronegative than fluorine, assume that the central atom is carbon. We have to place the least electronegative atom at the center. Lewis Dot Structure of Electron Groups Bond angles 3-D Sketch Name of Electron Geometry Name of Molecular Shape Name of Compound PART II CF4 32 v. So the total electron pairs = 32 ÷ 2 = 16 Total electron pairs = total valence electrons ÷ 2 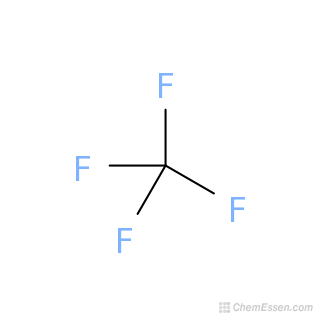
And when we divide this value by two, we get the value of total electron pairs. Learn how to find: Carbon valence electrons and Fluorine valence electrons Predict the molecular geometry of each molecule. The molecular geometry can be described as a trigonal planar arrangement with one vertex missing. Valence electrons of one carbon atom = 4 × 1 = 4 Valence electrons of four fluorine atoms = 7 × 4 = 28Īnd the total valence electrons = 4 + 28 = 32 D With two nuclei around the central atom and one lone pair of electrons, the molecular geometry of SnCl 2 is bent, like SO 2, but with a ClSnCl bond angle of 95. Since CF 4 has one carbon atom and four fluorine atoms, so… Hence, carbon has four valence electrons and fluorine has seven valence electrons. According to the VSEPR theory, if the molecule has an AX4 generic formula, the molecular geometry and electron geometry will both be tetrahedral. In the periodic table, carbon lies in group 14, and fluorine lies in group 17.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
